What Reagent Would Be Used to Reduce an Amide to an Amine?
* Lithium aluminium hydride, LiAlHfour, also abbreviated as LAH, is a reducing agent normally employed in modernistic organic synthesis.
* It is a nucleophilic reducing agent, best used to reduce polar multiple bonds like C=O.
* LiAlHiv reagent can reduce aldehydes to primary alcohols, ketones to secondary alcohols, carboxylic acids and esters to main alcohols, amides and nitriles to amines, epoxides to alcohols and lactones to diols.
* Lithium aluminium hydride, LAH reagent cannot reduce an isolated not-polar multiple bond like C=C. However, the double or triple bonds in conjugation with the polar multiple bonds can exist reduced.
* LiAlH4 is a powerful reducing agent compared to sodium borohydride, NaBHiv, since the Al-H bond is weaker and thus less stable than B-H bond.
1) Structure of Lithium aluminium hydride
2) Preparation
3) Properties
4) Machinery of Lithium aluminium hydride - LiAlHiv reduction
five) Applications of LiAlH4 in organic synthesis
Construction of Lithium aluminium hydride - LiAlH4
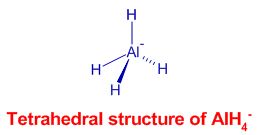
There is a tetrahedral arrangement of hydrogens around Al3+ in aluminium hydride, AlHiv - ion. It is formed by coordination of hydride, H- ions to Althree+ ion. The hybridization in cardinal Al is sp3.
Preparation of LiAlHfour
LiAlHfour is prepared by the reaction between lithium hydride and aluminium chloride.

Properties of LiAlH4 , Reaction conditions & Workup
* Lithium aluminium hydride, LAH is a white solid but the commercial samples are unremarkably gray due to presence of impurities.
* Information technology reacts violently with water by producing hydrogen gas. Hence it should non exist exposed to moisture and the reactions are performed in inert and dry temper.

* The reduction reaction employing LiAlH4 as reducing agent must be carried out in anhydrous non protic solvents like diethyl ether, THF etc. It is highly soluble in diethyl ether. Notwithstanding information technology may spontaneously decompose in it due to presence of catalytic impurities. Therefore the preferred solvent for LAH is THF despite the depression solubility.
* The reactions are usually performed with excess of LiAlH4. A pocket-sized amount of the reagent is added to the solvent to eliminate any wet present in the solvent.
Workup:
During the workup, the reaction mixture is initially chilled in an water ice bath and then the Lithium aluminium hydride is quenched by careful and very slow addition of ethyl acetate followed past the addition of methanol and so cold water.
Sometimes, the reagent is decomposed by adding undried solvent slowly and then dilute sulphuric acrid to the reaction mixture.
Mechanism OF REDUCTION BY LITHIUM ALUMINIUM HYDRIDE, LiAlH4
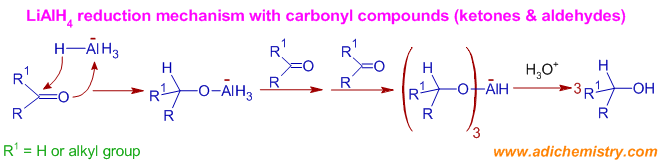
* The reduction of a carbonyl group by LiAlH4 is initiated past the assail of nucleophilic hydride ion on the carbonyl carbon to give a tetrahedral intermediate.
* LiAlH4 is a nucleophilic reducing agent since the hydride transfer to the carbonyl carbon occurs prior to the coordination to the carbonyl oxygen. It reacts faster with electron deficient carbonyl groups. The reactivity of carbonyl compounds with this reagent follows the gild:
Aldehydes > Ketones > ester > amide > carboxylic acid
* The steps involved in the reduction of various functional groups are shown beneath:
Machinery of Reduction of Aldehydes or Ketones to 10 or 20 alcohols: Initially, a hydride ion is transferred onto the carbonyl carbon and the oxygen atom coordinates to the remaining aluminium hydride species to furnish an alkoxytrihydroaluminate ion, which tin can reduce the next carbonyl molecule. Thus 3 of the hydride ions are used up in reduction.
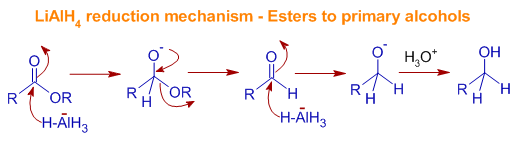
Machinery of Reduction of Esters to 10 alcohols past LiAlH4:The ester is get-go converted to aldehyde which is farther reduced to primary alcohol.
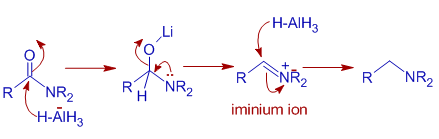
Mechanism of Reduction of Amides to amines: Amides are converted to amines. The LAH reduction machinery is slightly different from that depicted for esters. In iminium ion is formed during the reaction since nitrogen atom is relatively a proficient donor than oxygen cantlet.
Mechanism of Reduction of nitriles to primary amines by LiAlH4: Initially, the polar CN bond is added with LAH such that the negatively charged hydride makes bond with carbon. It is followed past subsequent transfer of hydride from AlH3 - group. Final protic workup generates amine group.
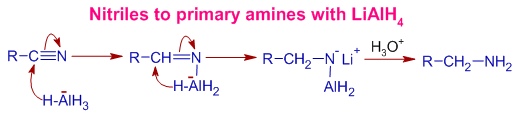
APPLICATIONS OF LiAlH4 IN ORGANIC SYNTHESIS
The summary nautical chart of applications of LiAlH4 in the reduction of different types of functional groups.
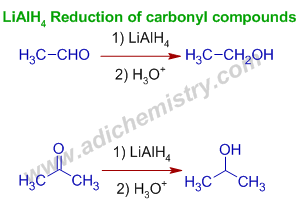
one) Reduction of carbonyl compounds using LiAlHfour : The aldehydes or ketones are reduced past LiAlHiv to the corresponding primary or secondary alcohols respectively.
Due east.g. Acetaldehyde is reduced to ethyl alcohol and acetone is reduced to isopropyl alcohol.
* LiAlH4 does not touch on the isolated carbon-carbon double or triple bonds.
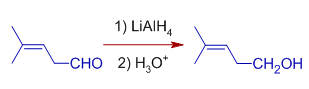
*Even so, the double bonds in conjugation at α,β positions of carbonyl group may also be reduced past Lithium aluminium hydride depending on the reaction atmospheric condition.
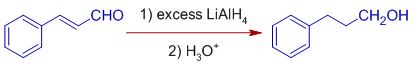
E.k. Cinnamaldehyde is reduced to Hydrocinnamyl booze when reduced with excess of LiAlH4 (roughly more than than ii equivalents) by normal improver method. In this method, a solution of cinnamaldehyde is added to the solution of lithium aluminium hydride. Both the double bail and carbonyl group are reduced.
Whereas, Cinnamaldehyde is reduced to Cinnamyl alcohol with one equivalent of LiAlHfour in inverse improver method. In this method, the solution of LiAlHiv is added to the solution of Cinnamaldehyde. Just the carbonly group is reduced to alcohol.
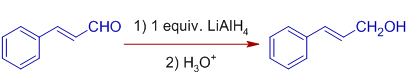
Stereochemistry:
The axial assault of hydride ion is preferred over the equatorial attack in case of cyclic systems. For case, 4-t-butylcyclohexanone yields more than xc% of trans-iv-t-butylcyclohexanol when reduced with Lithium aluminium hydride.
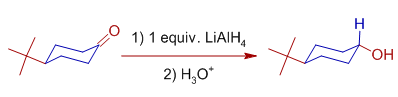
The plausible explanation for this beliefs is: the -OH group prefers the equatorial position to avert the interactions with other axial hydrogens. i.e., Information technology is not the arroyo of hydride ion but the orientation of -OH group which decides the terminal stereochemistry.
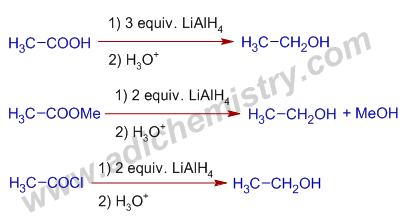
2) The carboxylic acids, esters and acrid halides are reduced to respective primary alcohols by Lithium aluminium hydride.
E.chiliad. The reduction of Acetic acrid, methyl acetate and acetyl chloride by LiAlH4 furnish the same ethyl alcohol.
three) The amides are reduced to amines by Lithium aluminium hydride, LiAlHfour. Specially this method is used to go secondary amines.
Eastward.g. Diethyl amine tin can be prepared starting from acetyl chloride every bit follows:
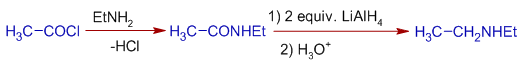
4) The nitriles are reduced to primary amines by LiAlHfour.
Due east.g. Acetonitrile is reduced to ethyl amine past LiAlH4.
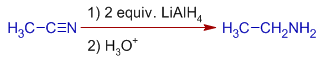
v) Lithium aluminium hydride reduces the oxiranes (epoxides) to alcohols. The mechanism involves hydride set on occurs at less hindered side of the epoxide.
E.thousand. ii-methyloxirane gives two-propanol predominantly.
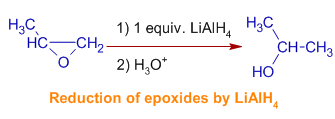
In case of cyclohexene epoxides, the centric alcohols are formed preferentially.
E.g.
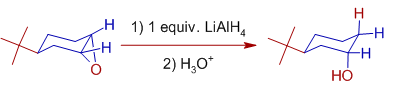
6) The lactones are reduced to α,ω-diols by LiAlHfour.
E.thousand.
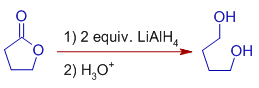
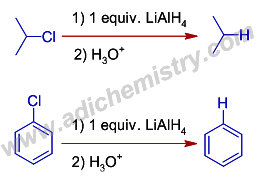
7) The haloalkanes and haloarenes are reduced to corresponding hydrocarbons by Lithium aluminium hydride.
TEST YOUR UNDERSTANDING - Lithium Aluminium Hydride Reagent
ane) What happens to the pH of water when LiAlH4 is is added to information technology?
Answer:
The H-, hydride ions can react violently with h2o to liberate hydrogen gas and the solution becomes alkaline, now containing LiOH and Al(OH)3. Thus pH is increased. It will be greater than seven. The reaction of Lithium aluminium hydride, LiAlH4 with h2o is shown below.
LiAlHfour + 4H2O -------> LiOH + Al(OH)iii + 4Hii
Information technology is strongly bones and hence can non only react with water but as well with protic solvents like methanol.
two) Why do we quench the reaction mixture with methanol after reduction with Lithium aluminium hydride?
Respond:
Just think.......what does LiAlHiv do with protic solvents? It can react with methanol in the same way every bit information technology reacts with water simply less violently since the O-H bond in methanol is less polar.
LiAlH4 + 4MeOH -------> LiOMe + Al(OMe)iii + 4Hii
Hence methyl alcohol is used in the quenching of LAH during workup. It is ameliorate to quench in cold weather condition.
LITHIUM ALUMINIUM HYDRIDE - LiAlHiv REDUCTION- MCQ - IIT JEE - NEET - JAM - GATE - CSIR Net & Fix EXAMS
1) The near appropriate reagent to convert RCOOEt -----> RCHiiOH is: (GATE 1997)
A) LiAlH4
B) NaBH4
C) Hii/Pd–C
D) Li/NH3(liq)
2) ethyl ethanoate on reduction with LiAlH4 gives
A) Ethanol
B) Diethyl ether
C) Ethane
D) Acetic acid and ethyl booze
3) When acetyl chloride is reduced with LiAlH4, the production formed is :
A) Ethanoic acrid
B) Ethyl chloride
C) Ethyl alcohol
D) Ethane
four) The reduction product of N-ethylpropanamide with LiAlH4 is :
A) Diethylamine
B) 1-propanamine
C) N-ethylpropan-ane-amine
D) North-ethylpropan-one-amine
5) The product formed when cyclopentanecarbaldehyde is reduced with LAH is :
A) cyclopentane
B) methylcyclopentane
C) ethylcyclopentane
D) cyclopentylmethanol
vi) Reduction of i-methylpyrrolidin-2-i with two equivalents of lithium aluminium hydride will give :
A) 1-methylpyrrolidine
B) 1-methylpyrrolidin-ii-ol
C) pyrrolidin-two-ol
D) pyrrolidin-2-1
seven) The reagent that can be used when four-methoxybenzoic acid is reduced with LAH is :
A) Diethyl ether
B) Ethyl alcohol
C) THF
D) Water
E) Both A & C can be used
KEY & Caption
1) A
2) A
3) C
4) C
5) D
half-dozen) A
seven) Eastward
Source: https://www.adichemistry.com/organic/organicreagents/lah/lithium-aluminium-hydride-1.html
0 Response to "What Reagent Would Be Used to Reduce an Amide to an Amine?"
Post a Comment